Patient Randomisation for
Simple Phase 1 Studies
Prancer RTSMⓇ Rand-Only offers simplified patient tracking for studies that require randomisation where inventory management is not a priority.
Alleviate the burden on sites to manually perform randomisation.
Prancer RTSMⓇ Rand-Only randomises into active or placebo treatment arms based on your study protocol rather than relying on traditional methods of opening envelopes to determine treatment.

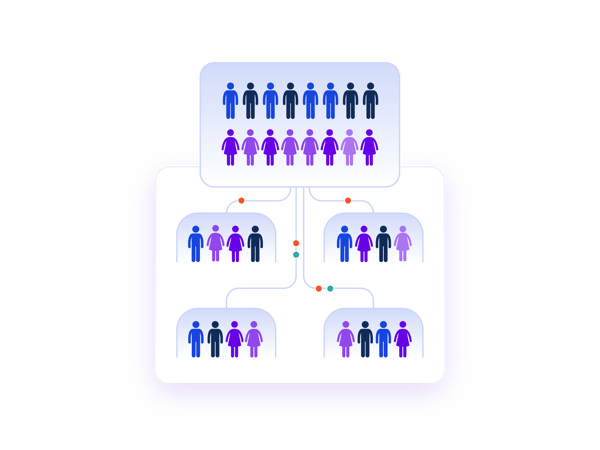
Centralise and simplify cohort assignments.
You may have plenty of supply and no worries about site stock. But, managing which patients are enrolled into which cohort across multiple sites takes a lot of manual effort. Leverage Prancer RTSM® Rand-Only to control cohort assignments and communication with your sites. You'll have greater transparency with critical investigators and a detailed audit trail to record how your study was conducted.
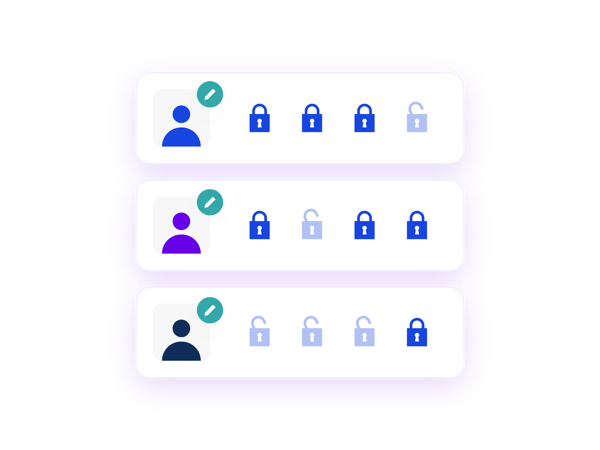
An ideal solution for blinded studies with unblinded pharmacists.
Prancer RTSMⓇ Rand-Only enables blinded site users to register patients and unblinded pharmacists to see the assigned treatment. Randomisation can be performed by blinded or unblinded site users with complete confidence that the study blind will be maintained.
Streamline Clinical Trial Management
With simple studies only requiring randomisation, Prancer RTSMⓇ Rand-Only helps reduce manual processes, improve efficiency, and ensures adherence to the study protocol.
Efficient Implementation
Due to its simplicity, Prancer RTSM® Rand-Only requires few design review sessions to finalise your specification. Validation and UAT focus on the simpler patient flow, allowing your study to go live faster.
Seamless Integrations
Allows for simple integrations with tools you already use.
Improved Accuracy
Avoid misrandomisation with automated processes.
Integrations Made Simple
Integrations shouldn’t hinder choosing the best-in-class systems for your clinical trial. Modern technology simplifies the integration process no matter what systems you are using.
RTSM solutions for any trial complexity.
Our randomisation and trial supply solutions can easily be configured to include only the needed functionality and are built to scale with you as your clinical programme progresses.
Prancer RTSM®
100% configurable and flexible solution with robust features and functionality to meet the needs of any trial.
Learn MorePrancer RTSM® Lite
Ideal for simple studies for improved accuracy over manual spreadsheets.
Learn MorePrancer RTSM® Platform
Management of Investigator Sponsored Trial (IST) supply tracking and oversight.
Learn More